New vaccines and companion diagnostics for infectious diseases
Together with the Medical University of Vienna with the team of Univ. Prof. Dr. Rudolf Valenta from the Center for Pathophysiology, Infectiology and Immunology Viravaxx AG has developed completely new concepts for preventive and therapeutic vaccines and companion diagnostics for infectious diseases. The specific approach of Viravaxx is to analyze immune responses in infected patients in order to learn how immunity can be improved. Based on this knowledge Viravaxx engineers novel and sophisticated immunogens to create preventive and therapeutic vaccines.
One focus is a vaccine against hepatitis B infections. Chronic hepatitis B (HBV) is a serious viral disease associated with inflammation of the liver. According to World Health Organization (WHO) estimations, there are currently more than 250 million sufferers worldwide. Treatment of chronic HBV usually includes nucleoside and nucleotide analogues or interferon therapy. However, these forms of treatment have many side-effects and do not have long-lasting protective effect once the medication intake is stopped. Current vaccinations to protect against HBV based on the S protein, a surface protein of HBV, are not effective in 10 – 20% of cases.
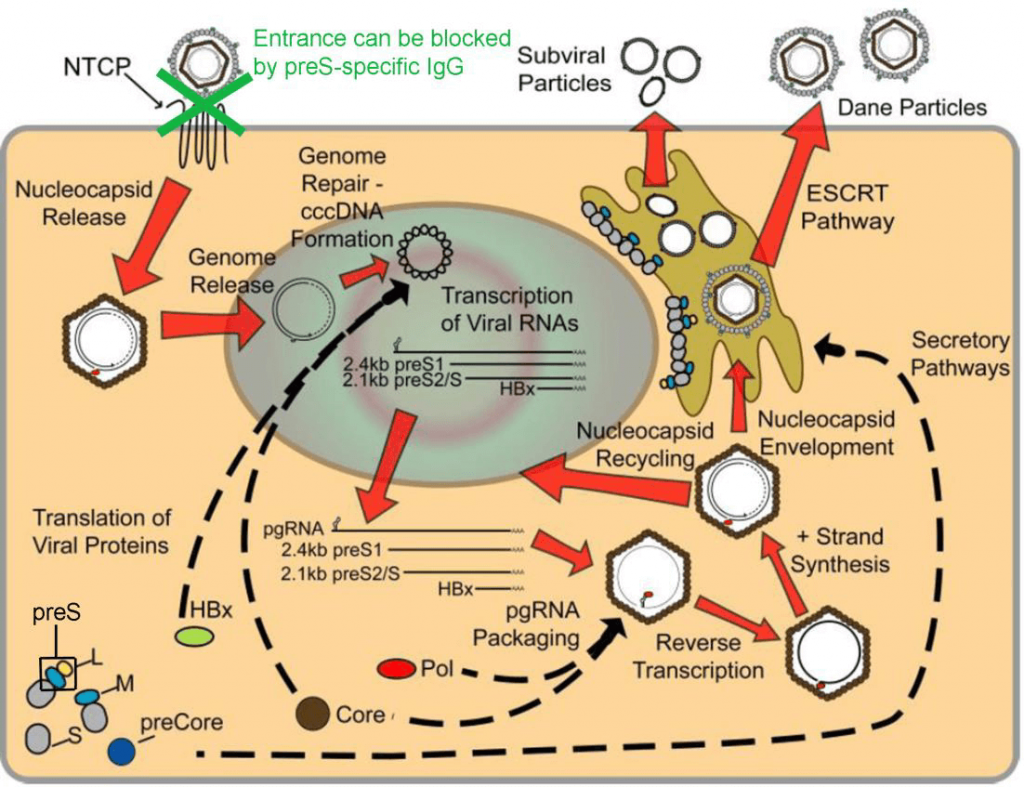
The newly developed vaccine VVX001 avoids these disadvantages and is a step towards the therapeutic as well as prophylactic use of vaccination against chronic hepatitis B. This vaccine (VVX001) contains preS, another surface protein which mediates the attachment of HBV to the human receptor NTCP on hepatocytes. The vaccine has already been tested successfully in clinical studies where a robust induction of preS-specific antibodies directed to the HBV attachment site (NTCP) and capable to block HBV infection in vitro was shown (see references 1 and 2).
Thus preS-specific antibodies have the potential to protect against HBV-infection in non-responders to HBsAg-based vaccines and to treat chronically HBV-infected patients by inhibiting binding of HBV to its receptor NTCP on liver cells (see modified Figure from Lamontage RJ et al., Hepatoma Research 2016 below).
A clinical study investigating its effect in HBV-infected patients as well as non-responders to HBsAg-based vaccines has been completed (ClinicalTrials.gov Identifier: NCT03625934). The results demonstrate an excellent safety profile of VVX001, the potential to provide protection in non-reponders and its ability to achieve functional cure in patients chronically infected with HBV by inducing preS-specific antibody and T cell responses.
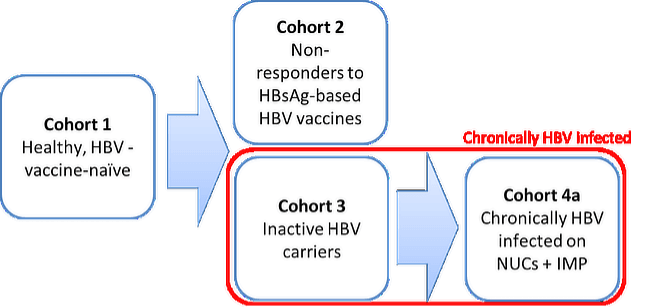
The primary endpoint, significant elicitation of PreS specific IgG antibodies in actively treated subjects compared to placebo was achieved in all four cohorts. (p<0.001 in all cohorts and overall).
These antibodies were shown to inhibit HBV virus spreading in HepG2-NTCP cells in a cell culture model, which validates the virus neutralization capacity of the vaccination. HBV- neutralizing antibodies could be induced in non-responders to conventional vaccination. Encouragingly, more than half of the chronically infected patients in the study had a >50% reduction of serum HbSAg at 8 months after the end of treatment, which points to the potential of achieving a functional cure with a finite course of treatment with VVX001. On prolonged follow-up, a further decrease in some patients was observed.
Vaccination with VVX001 was safe and very well tolerated. Most importantly, direct antivirals could be safely stopped in most chronically infected patients after three VVX001 injections without a virus flare.
A second project is a vaccine to protect against SARS-CoV-2. The antigen-based vaccine targets the receptor binding domains (RBD) of the SARS-CoV-2 virus and induce a robust and uniform RBD-specific IgG antibody response in animal models and in human tests. This antibody response prevents the virus from docking on to and entering in the body’s cells, so that infection cannot occur. The preclinical data for this vaccine indicates that it is effective against all SARS-CoV-2 variants known to date, including omicron – even in those who have not yet built up any immunity as a result of vaccination (non-responders) (see reference 4).
The SARS-CoV-2 subunit vaccine (PreS-RBD) developed by Viravaxx in collaboration with MedUni Vienna is based on a structurally folded fusion protein consisting of two receptor binding domains (RBD) of the SARS-CoV-2 virus and the PreS antigen from hepatitis B virus, which serve as immunological carriers for each other, thereby strengthening the immune response. So this vaccine can be used as a combination of coronavirus vaccine and hepatitis B vaccine (see reference 4).
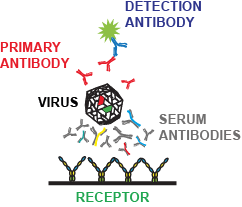
Viravaxx together with Medical University of Vienna has also developed a test, which can indicate not only if and how much antibodies somebody has, but also inform about the quality of the antibody response and its ability to neutralize the virus. This allows to check if an infected or vaccinated person has immunity which can protect. This is especially important in the fight against COVID-19 (see reference 5-8).
A third development is a vaccine and companion diagnostics for the Respiratory syncytial virus (RSV). RSV causes severe symptoms in infants and elderly people and alone or with rhinovirus (RV) induces severe exacerbations of chronic respiratory diseases such as asthma, COPD (Chronic Obstructive Pulmonary Disease) and ACOS (Asthma and COPD Overlap Syndrome). No active vaccine is available on the market until now.
The development of high- resolution diagnostic tests for RV and RSV infections allows a stratification and personalized treatment of patients with asthma, (treatment of allergic asthma or allergen- and virus-triggered asthma or only virus-triggered asthma) opening the doors for personalized medicine in severe respiratory diseases (see references 9 and 10).
In addition to this Viravaxx AG holds patents and IP for diagnostics for HIV and RV (see references 11-13).


Opportunity
Acquisition of one or more vaccines and diagnostics in the form of an asset deal or share deal. If you are interested, you will get access to our data room and Univ. Prof. Dr. Rudolf Valenta and his team are ready for detailed presentations after signing of a CDA.
References
1) Cornelius C, Schöneweis K, Georgi F, Weber M, Niederberger V, Zieglmayer P, Niespodziana K, Trauner M,Hofer H, Urban S, Valenta R. Immunotherapy With the PreS-based Grass Pollen Allergy Vaccine BM32 Induces Antibody Responses Protecting Against Hepatitis B Infection. EBioMedicine. 2016 Sep;11:58-67.doi: 10.1016/j.ebiom.2016.07.023.
2) Tulaeva I, Cornelius C, Zieglmayer P, Zieglmayer R, Schmutz R, Lemell P, Weber M, Focke-Tejkl M, KaraulovA, Henning R, Valenta R. Quantification, epitope mapping and genotype cross-reactivity of hepatitis B preS-specific antibodies in subjects vaccinated with different dosage regimens of BM32. EBioMedicine. 2020 Sep;59:102953. doi: 10.1016/j.ebiom.2020.102953.
3) Steindl-Munda PE, Tulaeva I, Aberle JH, Kozbial K, Holzmann H, Wiedermann-Schmidt U, Brunar H, TraunerMH, Valenta R and Ferenci P. VVX001 induces robust preS-specific immunity in patients chronicallyinfected with hepatitis B. Hepatology 2021;74 (Suppl.1):52-523A. doi: 10.1002/hep.32188
4) Gattinger P, Kratzer B, Tulaeva I, Niespodziana K, Ohradanova-Repic A, Gebetsberger L, Borochova K,Garner-Spitzer E, Trapin D, Hofer G, Keller W, Baumgartner I, Tancevski I, Khaitov M, Karaulov A, StockingerH, Wiedermann U, Pickl WF, Valenta R. Vaccine based on folded RBD-PreS fusion protein with potential to induce sterilizing immunity to SARS-CoV-2 variants. Allergy. 2022 Mar 31. doi: 10.1111/all.15305.
5) Gattinger P, Borochova K, Dorofeeva Y, Henning R, Kiss R, Kratzer B, Mühl B, Perkmann T, Trapin D, TrellaM, Ettel P, Tulaeva I, Pickl WF, Valenta R. Antibodies in serum of convalescent patients following mildCOVID-19 do not always prevent virus-receptor binding. Allergy. 2021 Mar;76(3):878-883. doi: 10.1111/all.14523.
6) Gattinger P, Niespodziana K, Stiasny K, Sahanic S, Tulaeva I, Borochova K, Dorofeeva Y, Schlederer T,Sonnweber T, Hofer G, Kiss R, Kratzer B, Trapin D, Tauber PA, Rottal A, Körmöczi U, Feichter M, Weber M,Focke-Tejkl M, Löffler-Ragg J, Mühl B, Kropfmüller A, Keller W, Stolz F, Henning R, Tancevski I,Puchhammer-Stöckl E, Pickl WF, Valenta R. Neutralization of SARS-CoV-2 requires antibodies againstconformational receptor-binding domain epitopes. Allergy. 2022 Jan;77(1):230-242. doi:10.1111/all.15066.
7) Gattinger P, Tulaeva I, Borochova K, Kratzer B, Trapin D, Kropfmüller A, Pickl WF, Valenta R. Omicron: ASARS-CoV-2 variant of real concern. Allergy. 2022 May;77(5):1616-1620. doi: 10.1111/all.15264.
8) Mair MJ, Mitterer M, Gattinger P, Berger JM, Trutschnig W, Bathke AC, Gansterer M, Berghoff AS, LaengleS, Gottmann L, Buratti T, Haslacher H, Lamm WW, Raderer M, Tobudic S, Fuereder T, Valenta R, Fong D,Preusser M. Cancer Cell. 2022 Apr 12:S1535-6108(22)00165-9. doi: 10.1016/j.ccell.2022.04.003.
9) Niespodziana K, Borochova K, Pazderova P, Schlederer T, Astafyeva N, Baranovskaya T, Barbouche MR, Beltyukov E, Berger A, Borzova E, Bousquet J, Bumbacea RS, Bychkovskaya S, Caraballo L, Chung KF,Custovic A, Docena G, Eiwegger T, Evsegneeva I, Emelyanov A, Errhalt P, Fassakhov R, Fayzullina R, Fedenko E, Fomina D, Gao Z, Giavina-Bianchi P, Gotua M, Greber-Platzer S, Hedlin G, Ilina N, Ispayeva Z, Idzko M,Johnston SL, Kalayci Ö, Karaulov A, Karsonova A, Khaitov M, Kovzel E, Kowalski ML, Kudlay D, Levin M,Makarova S, Matricardi PM, Nadeau KC, Namazova-Baranova L, Naumova O, Nazarenko O, O’Byrne PM, Osier F, Pampura AN, Panaitescu C, Papadopoulos NG, Park HS, Pawankar R, Pohl W, Renz H, Riabova K, Sampath V, Sekerel BE, Sibanda E, Siroux V, Sizyakina LP, Sun JL, Szepfalusi Z, Umanets T, Van Bever HPS,van Hage M, Vasileva M, von Mutius E, Wang JY, Wong GWK, Zaikov S, Zidarn M, Valenta R. Towardpersonalization of asthma treatment according to trigger factors. J Allergy Clin Immunol. 2020Jun;145(6):1529-1534. doi: 10.1016/j.jaci.2020.02.001.
10) Borochova K, Niespodziana K, Stenberg Hammar K, van Hage M, Hedlin G, Söderhäll C, Focke-Tejkl M,Valenta R. Features of the Human Antibody Response against the Respiratory Syncytial Virus SurfaceGlycoprotein G. Vaccines (Basel). 2020 Jun 25;8(2):337. doi: 10.3390/vaccines8020337.
11) Niespodziana K, Stenberg-Hammar K, Megremis S, Cabauatan CR, Napora-Wijata K, Vacal PC, Gallerano D, Lupinek C, Ebner D, Schlederer T, Harwanegg C, Söderhäll C, van Hage M, Hedlin G, Papadopoulos NG,Valenta R. PreDicta chip-based high resolution diagnosis of rhinovirus-induced wheeze. Nat Commun. 2018Jun 18;9(1):2382. doi: 10.1038/s41467-018-04591-0.
12) Pazderova P, Waltl EE, Niederberger-Leppin V, Flicker S, Valenta R, Niespodziana K. ELISA-Based Assay forStudying Major and Minor Group Rhinovirus-Receptor Interactions. Vaccines (Basel). 2020 Jun 18;8(2):315.doi: 10.3390/vaccines8020315.
13) Gallerano D, Wollmann E, Lupinek C, Schlederer T, Ebner D, Harwanegg C, Niespodziana K, Schmetterer K,Pickl W, Puchhammer-Stöckl E, Sibanda E, Valenta R. HIV microarray for the mapping and characterizationof HIV-specific antibody responses. Lab Chip. 2015 Mar 21;15(6):1574-89. doi: 10.1039/c4lc01510j.
